The common earthball, Scleroderma citrinum Pers. The white plates of the trama may be seen, marbled in the black glebal spore mass.
Classification
Kingdom Fungi
Phylum Basidiomycota
Class Basidiomycetes
Order Boletales
Family Sclerodermataceae
Genus Scleroderma
Synonyms
Lycoperdon aurantium sensu auct.
fide Checklist of Basidiomycota of Great Britain and Ireland (2005)
Scleroderma aurantiacum sensu Carleton Rea (1922), Ramsbottom (1953), non Linnaeus (Sp. Pl., 1753)
fide Checklist of Basidiomycota of Great Britain and Ireland (2005)
Scleroderma aurantium sensu auct.
fide Checklist of Basidiomycota of Great Britain and Ireland (2005)
Scleroderma vulgare Hornem.
Fl. Danic.: tab. 1969, fig. 2 (1829)
Common names
Common earthball
Poison pigskin puffball
Kartoffelbovist
Scléroderme vulgaire
Scléroderme orangé
Description
Fruit body: 2-12 cm in diameter; roughly spherical in shape; the surface hard and scaly, yellowish to yellow-brown, often cracked; the peridium is whitish when sliced but blushing pinkish; mycelial strands may be found attaching the puffball to the ground. In maturity, the peridium breaks open to release the spores.
Gleba: thick and white initially, later purple-black from the center outwards, marbled by white tramal plates; eventually blackish to brownish and dust-like.
Spores: brown, globose, reticulate, with spines up to 1.5 µm long; 8-13 µm diameter.
Habitat: this species is mycorrhizal with hardwoods and conifers; often found in mossy areas, especially on sandy soils; growing alone, scattered, or gregariously; widely distributed; summer and fall.
Edibility: inedible/poisonous; ingestion possibly causing gastric disturbances in some individuals.
Chemical reactions: the surface becomes dark reddish in color with the application of KOH.
This species may be parasitized by Boletus parasiticus.

My name is Austin Collins.
I've dedicated my life to Mushrooms.
I believe Mushrooms are the best kept secret when it comes to health and well being.
For that reason, I would like to share a company with you that in my opinion makes the best mushroom products on the market.
The company is called Noomadic Herbals, my favorite supplement they make is called "Mushroom Total".
I take their products every day and they have helped me think better and have more energy. Give them a try.
-Austin
Bioactive compounds
A number of bioactive compounds have been isolated, and some characterised from S. citrinum:
- the sterol 23ξ-hydroxylanosterol was isolated from the peridium (Entwistl and Pratt, 1968; 1969)
- the lanostane-type triterpenoid, (20S,22S,23E)-22-o-acetyl-25-hydroxylanosta-8,23(E)-dien-3-one, had significant antiviral activity against Herpes simplex type 1
- methyl 4,4′-dimethoxyvulpinate and two derivatives-dibromo and acetate-showed antibacterial activity towards the pathogen Mycobacterium tuberculosis; the derivatives were also cytotoxic to the NCI-H187 cell line
- 4,4′-dimethoxyvulpinic acid (Kanokmedhakul et al., 2003)
Pulvinic acid dimers have also been isolated from Scleroderma citrinum, including norbadione A, badione A and sclerocitrin. The latter compound is a pigment that gives the fungus its yellow color, and is present in relatively high amounts-400 mg/kg fungus (Winner et al., 2004). This pigment is also found in the peppery bolete, Chalciporus piperatus.
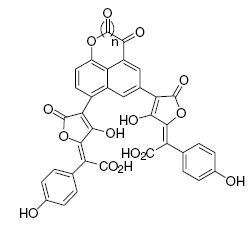
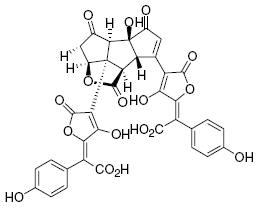
Chemical structure of the yellow pigment sclerocitrin.
Links
References
Entwistl N, Pratt AD.
23ξ-hydroxy-lanosterol – a new triterpene fungal metabolite of basidiomycete Scleroderma aurantium Pers.
Tetrahedron. 1968 24(10):3949-53.
Entwistl N, Pratt AD.
Determination of absolute configuration at C23 in 23-hydroxylanosterol – a triterpene fungal metabolite of basidiomycete Scleroderma aurantium Pers and its C23 epimer.
Tetrahedron. 1969 25(7):1449-51.
Garrido N.
Notes on Scleroderma citrinum Pers (mycota-Gasteromycetes) in Pinus radiata forests in Chile.
Nova Hedwigia. 1984 40(1-4):511-6.
Kannan K, Natarajan K.
Pure culture synthesis of Pinus patula ectomycorrhizae with Scleroderma citrinum.
Current Science. 1987 56(20):1066-8.
Kanokmedhakul S, Kanokmedhakul K, Prajuabsuk T, Soytong K, Kongsaeree P, Suksamrarn A.
A bioactive triterpenoid and vulpinic acid derivatives from the mushroom Scleroderma citrinum.
Planta Med. 2003 69(6):568-71.
Pubmed
Natarajan K, Kannan K.
On the occurrence of Scleroderma citrinum in India.
Current Science. 1979 48(24):1086-7.
View PDF
Nikonorow M, Grzybowska J, Karkocha I.
[Domestic poisonous mushrooms. I. Chemical examination of Scleroderma aurantium].
Rocz Panstw Zakl Hig. 1967 18(3):277-81.
Pubmed
Vrkoc J, Budesinsky M, Dolejs L.
Constituents of Basidiomycete Scleroderma aurantium.
Phytochemistry. 1976 15(11):1782-4.
Winner M, Gimenez A, Schmidt H, Sontag B, Steffan B, Steglich W.
Unusual pulvinic acid dimers from the common fungi Scleroderma citrinum (common earthball) and Chalciporus piperatus (peppery bolete).
Angew Chem Int Ed Engl. 2004 43(14):1883-6.
Pubmed



Hi there – hoping you can help me in identifying what I think is a mushroom. I live in Tucson, AZ in a well populated neighborhood near the University of Arizona. I’ve found several 2″-3″ dia brown mounds growing from the earth, often in a cluster. Brown dust or spores are released when I’m watering the surrounding plants and accidentally splash them. I think they are toxic and I’d really like to remove the mounds. Can you please tell me the best way to to that safely as well as prevent future growth.
Thank you
I have an article specific to Arizona.