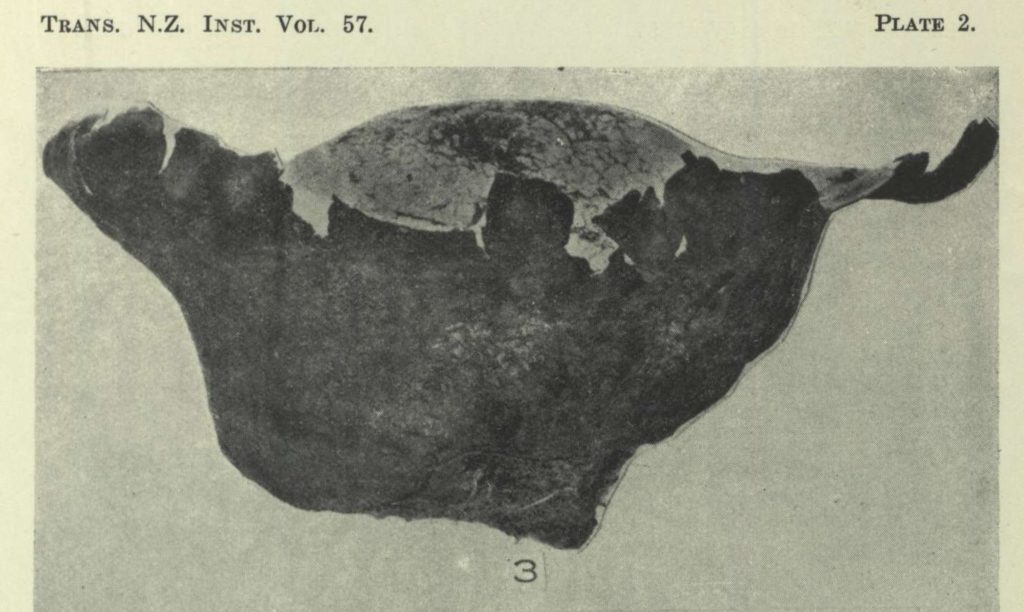
Sterile base; note the conspicuous diaphragm
Classification
Kingdom Fungi
Phylum Basidiomycota
Class Basidiomycetes
Order Agaricales
Family Lycoperdaceae
Genus Calvatia
Synonyms
Bovista lilacina Mont. & Berk.
London Journal of Botany 4: 64 (1845)
Globaria lilacina (Mont. & Berk.) Speg.
(1880)
Lycoperdon lilacinum (Mont. & Berk.) Speg.
Description
“Peridium up to 15 cm. diam., subglobose or pyriform, tapering abruptly into a large, well-developed, strongly crenulate rooting base; exoperidium smooth or more frequently floccose, often areolate, cream to bay brown, thin, fragile, fugacious; endoperidium brown, thin, fragile, breaking away irregularly from the apical portion; sterile base well developed, persistent, cellular at the periphery, hemicompact within, separated from the gleba by a prominent diaphragm.
Gleba some shade of purple, sometimes with a greyish tinge, at first compact, soon pulverulent; capillitium threads long, branched, septate, equal, pallid olivaceous.
Spores: globose, 5.5–7.5 mm. diam., occasionally apiculate; epispore strongly verrucose, violaceous.
Habitat: Solitary on the ground, usually in sandy areas.
Characterized by the prominent sterile base, conspicuous diaphragm, and especially by the strongly verrucose, violaceous spores. The peridium and gleba are fragile, consequently the sterile base is often the only portion of the plant collected; nevertheless even this can be determined readily owing to its structure. It is liable to confusion only with that of C. caelata, but may be separated by its peculiar, partly compact, partly cellular structure.
The peridium is usually stated to be smooth externally, but this is by no means a constant feature; on the contrary, collections frequently show the exterior to be floccose and even areolate.”
(Cunningham, 1925)

My name is Austin Collins.
I've dedicated my life to Mushrooms.
I believe Mushrooms are the best kept secret when it comes to health and well being.
For that reason, I would like to share a company with you that in my opinion makes the best mushroom products on the market.
The company is called Noomadic Herbals, my favorite supplement they make is called "Mushroom Total".
I take their products every day and they have helped me think better and have more energy. Give them a try.
-Austin
Medicinal properties
Antibacterial/antifungal activity
The structure of an antibacterial and antifungal compound isolated from the culture broth of Calvatia lilacina has been described on the basis of chemical and spectroscopic studies as p-carboxyphenylazoxycyanide (calvatic acid) (Gasco et al., 1974). The antimicrobial activity spectra of this compound was reported by Umezawa et al., (1975), who isolated the compound from a closely related species, Calvatia craniformis. Calvatic acid (see structure below) inhibited the growth of Gram-positive bacteria (3.12-6.25 µg/ml) and some Gram-negative bacteria, while showing no activity against yeasts and fungi at 100 µg/ml.
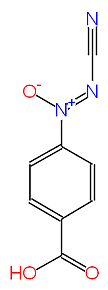
The exact stereoisomerism and position of the oxygen atom in the azoxycyano group were later confirmed by X-ray cystallography (Viterbo et al., 1975). A research group led by Gasco developed a synthetic route for making calvatic acid and also studied both the antimicrobial and antimitotic activities of several chemical analogues of this compound (Calvino et al., 1986).
The Umezawa et al. study (1975) reported that calvatic acid has antitumor activity: it inhibited the growth of Yoshida sarcoma in cell culture (IC50=1.56 µg/ml), and also showed growth inhibition of mouse leukemia 1210.
- This site describes a patent for large-scale production of calvatic acid.
Calvino R, Fruttero R, Gasco A.
Chemical and biological studies on calvatic acid and its analogues.
J. Antibiot. 1986 39(6):864-8.
Cunningham GH.
Lycoperdaceae of New Zealand.
Trans Proc Roy Soc New Zealand. 1925 57:190-217
Gasco A, Serafino A, Mortarin V, Menziani E, Bianco MA, Scurti JC.
An antibacterial and antifungal compound from Calvatia lilacina.
Tetra Lett. 1974 (38):3431-2.
Umezawa H, Takeuchi T, Iinuma H, Ishizuka M, Kurakata Y, Umeda Y, Nakanishi Y, Nakamura T, Obayashi A, Tanabe O.
A new antibiotic, calvatic acid.
J. Antibiot. 1975 38(1):87-90.
Viterbo D, Gasco A, Serafino A, Mortarini V.
p-Carboxyphenylazoxycyanide-dimethyl sulfoxide – antibacterial and antifungal compound from Calvatia lilacina.
Acta Crys Sec B-Struct Sci. 1975 31:2151-3.




Is there anything known about the edibility of this species when young?
Very little is known about Calvatia lilacina’s edibility.
I’m curious if this mushroom can be dehydrated and turned into a consumable powder form?
I’m in Georgia and the humid climate popped up a few big ones near my house.
Thanks
Technically you can do that with any mushroom.