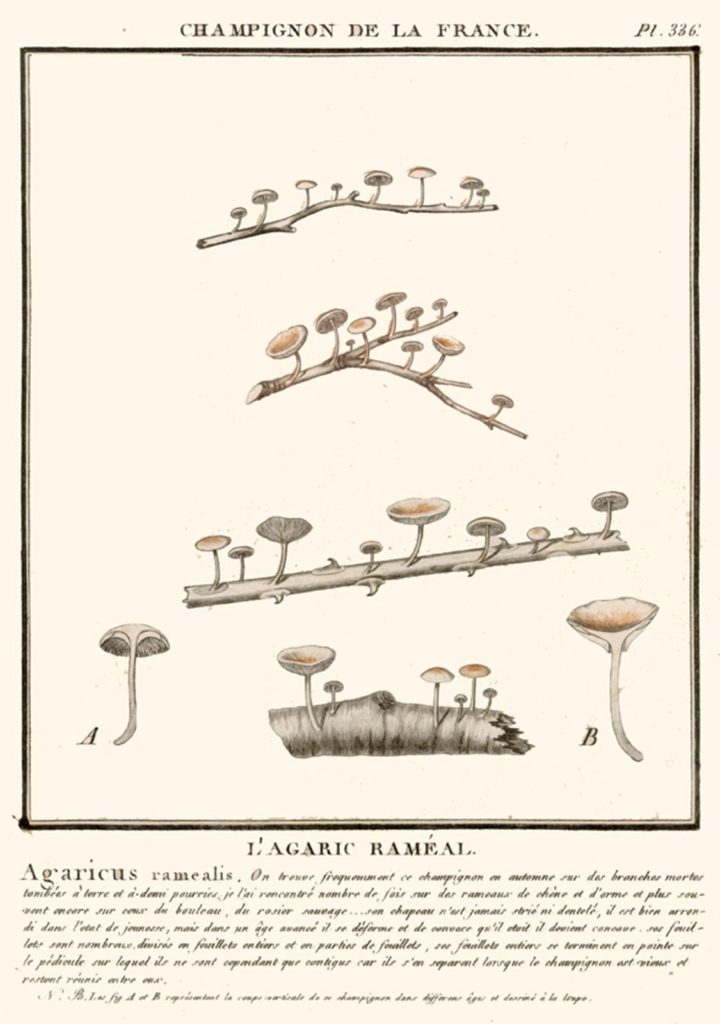
Picture source: Bulliard, P. 1791. Histoire des champignons de la France. I: 1-368.

Synonyms
Agaricus amadelphus Bull.
Agaricus ramealis Bull.
Collybiopsis ramealis (Bull.) Earle
Gymnopus ramealis (Bull.) Gray
Gymnopus ramealis (Bull.) J.L. Mata & R.H. Petersen
Marasmiellus amadelphus (Bull.) Singer
Marasmius amadelphus (Bull.) Fr.
Marasmius ramealis (Bull.) Fr.
Micromphale amadelphum (Bull.) Honrubia [as ‘amadelphus’]
Micromphale rameale (Bull.) Kühner [as ‘ramealis’]
Common names
Twig Parachute
Twig Mummy-cap
Pinwheel Marasmius
Ast-Schwindling (German)
Description
Cap: 3-10 mm across, convex then flattened or centrally depressed, whitish pink, often darker in the centre, membranous and often wrinkled.
Stem: 3-20 tall x 1 mm thick, scurfy, concolorous with cap, darkening towards the curved base.
Flesh: thin, concolorous.
Gills: distant, white or pinkish.
Spore print: white.
Spores: elongate, elliptical, 8.0-10 x 3-4 µm.
Habitat: on old stems.
Season: early summer to autumn. Frequent. Not edible. Found in Europe.

My name is Austin Collins.
I've dedicated my life to Mushrooms.
I believe Mushrooms are the best kept secret when it comes to health and well being.
For that reason, I would like to share a company with you that in my opinion makes the best mushroom products on the market.
The company is called Noomadic Herbals, my favorite supplement they make is called "Mushroom Total".
I take their products every day and they have helped me think better and have more energy. Give them a try.
-Austin
Bioactive compounds
Bendz described the isolation of the antibiotic marasin, shown in figure 1a (Bendz, 1959b).
A)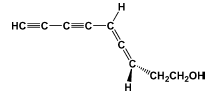 B)
B)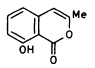 C)
C)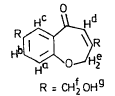
| Figure 1. Compounds found in Marasmiellus ramealis. A (-)-marasin. B 8-hydroxy-3-methylisocoumarin. C 3,7-bis(hydroxymethyl)-1-benzoxepin-5(2H)-one, an allyl aryl ether. |
Soon after (Bendz, 1960), other bioactive substances were identified, including (-)-nona-3,4-diene-6,8-diyne-l-ol. Furthermore, M. ramealis was also shown to produce several other closely related polyacetylenes and an iso-coumarin derivative, identified as 3-methyl-8-hydroxy-isocoumarin (figure 1b).
Using 14C-labeled precursors, the biosynthetic pathway for the production of the marasin was investigated, suggesting that the diyne-allene moeity in the polyacetylenic marasin is introduced enzymatically, via arrangement of an alkyltriyne moeity (Davies and Hodge, 2005).
The metabolite 3,7-bis(hydroxymethyl)-1-benzoxepin-5(2H)-one (figure 1c), has a benzoxepinone ring structure – at the time of discovery, unknown in natural product chemistry (Holroyde et al., 1976, 1978).
Later work would reveal the presence of the fungal metabolite mellein, as well as pyrrole-2-carboxylic acid and p-hydroxybenzoic acid (Jarrah and Thaller, 1983).
- This patent describes the production of 5-substituted picolinic acid derivatives by Marasmiellus species, including M. ramealis.
Bendz G.
8-Hydroxy-3-methylisocoumarin isolated from the culture medium of Marasmius ramealis.
Arkiv for Kemi. 1959a 14(6):511-8.
Bendz G.
Marasin, an antibiotic polyacetylene, isolated from the culture medium of Marasmius ramealis.
Arkiv for Kemi. 1959b 14(4):305-21.
Bendz G.
A study of the chemistry of some Marasmius species.
Arkiv for Kemi. 1960 15(2):131-48.
Bendz G, Wallmark G, Oblom K.
The antibiotic agent from Marasmius ramealis.
Nature. 1948 162(4106):61-2.
Davies DG, Hodge P.
Biosynthesis of the allene (-)-marasin in Marasmius ramealis.
Org Biomol Chem. 2005 3(9):1690-3.
Desjardin DE.
New and noteworthy marasmioid fungi from California.
Mycologia. 1987 79(1):123-34.
First page available online here
Holroyde JK, Orr AF, Thaller V.
3,7-Bis(hydroxymethyl)-1-benzoxepin-5(2h)-one, a novel metabolite from cultures of fungus Marasmiellus ramealis (Bull Ex Fr) Singer.
J Chem Soc-Chem Comm. 1976 (7):242-3.
Holroyde JK, Orr AF, Thaller V.
3,7-Bis(hydroxymethyl)-1-benzoxepin-5(2h)-one, a novel oxygen heterocyclic metabolite from cultures of the fungus Marasmiellus ramealis (Bull Ex Fr) Singer.
J Chem Soc-Perk Trans 1. 1978 (12):1490-3.
Jarrah MY, Thaller V.
Isolation and partial synthesis of 3-methoxycarbonyl- 7-formyl-1-benzoxepin-5(2h)-one the ester of a metabolite from shake cultures of the fungus Marasmiellus ramealis (Bull. Ex Fr.) Singer.
J Chem Soc-Perk Trans 1. 1983 (8):1719-21.