Dead man’s foot, Pisolithus tinctorius (Pers.) Coker and Couch. Note the brown spore deposit at the top of the picture.
Synonyms
Pisolithus arhizus (Scop.:Pers) Rauschert
Scleroderma tinctorium Pers.
Syn. meth. fung. (Göttingen) 1: 152 (1801)
Common names
Bohemian truffle
Dead man’s foot
Dyeball/dyemaker’s puffball
Horse dung fungus
Pea rock
Description
Fruiting body: initially globose to clavate, 4-12 cm diameter, 4-25 cm high, irregularly club-shaped, dull white, spotted olive-brown, yellowish brown to dingy brown with age.
Peridium: breaking open and disintegrating at the apex, releasing the brown powdery spores (see photo above). The gleba is composed of oval locules, pure white at first, becoming yellowish then dark brown and powdery when mature. Locules mature from the top down and more locules form near the base until almost the entire fruiting body is converted into a powdery mass.
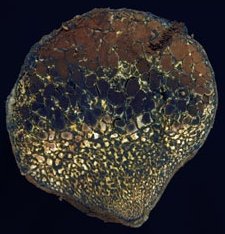
Odor: mild when young, but unpleasant with age.
Spore print: brown/cinnamon
Spores: globose, with long (up to 1 µm) spines, thick-walled, 7-12 µm.
Habitat: single to several, occasionally gregarious on ground under hardwoods and conifers; widely distributed; fruiting in spring, summer and fall during wet weather.
Edibility: inedible.
P. tinctorius forms mycorrhizal relationships with a variety of plants, especially Pinus and Eucalyptus species; the mushroom mycelium is routinely used to help initiate and establish new forests.
Dyemakers have used this mushroom when a brown color is desired.

My name is Austin Collins.
I've dedicated my life to Mushrooms.
I believe Mushrooms are the best kept secret when it comes to health and well being.
For that reason, I would like to share a company with you that in my opinion makes the best mushroom products on the market.
The company is called Noomadic Herbals, my favorite supplement they make is called "Mushroom Total".
I take their products every day and they have helped me think better and have more energy. Give them a try.
-Austin
Bioactive compounds
A number of triterpenes have been isolated from this species using successive column and preparative thin-layer chromatography techniques (Zamuner et al., 2005):
- 3β,22x,23x-trihydroxy-24-methyllanosta-8,24(28)-diene-31-al 22-acetate, a novel triterpene pisosteral
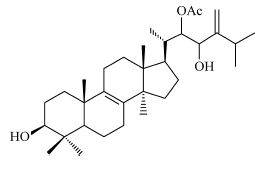
- 3β,22x,23x-trihydroxy-24-methylianosta-8,24(28)-diene 22-acetate (pisosterol, shown below)
- 3α,22x,23x-trihydroxy-24-methyllanosta-8,24(28)-diene 22-acetate (3-epi-pisosterol)
- 3β,22x,23x-trihydroxy-24-methyllanosta-8,24(28)-diene 23-acetate
- 3β,22x,23x-trihydroxy-24-ethyllanosta-8,24(28)-diene 22-acetate
- 3β,22x-dihydroxy-24-methyllanosta-8,24(28)-diene
- 3β,22x-dihydroxy-24-ethyllanosta-8,24(28)-diene
- 3β-hydroxylanosta-8,24-diene (lanosterol)
- 3β-hydroxylanosta-7(8),9,24-triene (agnosterol)
The first four listed triterpenes were isolated from a fungus growing in association with Eucalyptus trees, and the latter five from a mycelial culture of a strain that had been growing under Pinus taeda. These differences in triterpene composition from different P. tinctorius strains may suggest that the fungus is able to adapt and optimize its phytochemical output to establish the optimum mycorrhizal relationship with the host.
Medicinal properties
Antitumor effects
The triterpene pisosterol (chemical structure shown above) has been shown to have antitumor activity against seven tumor cell lines, especially leukemia and melanoma cells (IC50 of 1.55, 1.84 and 1.65 µg/ml for CEM, HL-60 and B16, respectively) (Montenegro et al., 2004). The compound has also been shown to induce a monocytic cell-like differentiation of the leukemia cell line HL-60 (Montenegro et al., 2007). Additionally, a recent in vivo study has corroborated the in vitro results. Montenegro et al. (2008) demonstrated that Sarcoma 180-bearing mice treated with pisosterol had significant tumor growth inhibition rates – 43.0% and 38.7% for mice treated with pisosterol at 10 or 100mg/m2, respectively. However, the liver and kidney suffered what the authors described as a ‘reversible’ damage.
Links
Tom Volk’s Fungus of the Month
Mushroom Expert
References
Montenegro RC, de Vasconcellos MC, Bezerra FS, Andrade-Neto M, Pessoa C, de Moraes MO, Costa-Lotufo LV.
Pisosterol induces monocytic differentiation in HL-60 cells.
Toxicol In Vitro. 2007 21(5):795-800.
Montenegro RC, Farias RAF, Pereira MRP, Alves APNN, Bezerra FS, Andrade-Neto M, Pessoa C, de Moraes MO, Costa-Lotufo LV.
Antitumor activity of pisosterol in mice bearing with S18 tumor.
Biol Pharm Bull. 2008 31(3):454-7.<
Montenegro RC, Jimenez PC, Farias RAF, Andrade-Neto M, Bezerra FS, Moraes MEA, de Moraes MO, Pessoa C, Costa-Lotufo LV.
Cytotoxic activity of pisosterol, a triterpene isolated from Pisolithus tinctorius (Mich.: Pers.) Coker & Couch, 1928.
Z Naturfors C. 2004 59(7-8):519-22.
Zamuner MLM, Cortez DAG, Dias BP, Lima MIS, Rodrigues E.
Lanostane triterpenes from the fungus Pisolithus tinctorius.
J Brazil Chem Soc. 2005 16(4):863-7.
Online PDF




What is the best way to apply this mycorrhizal to one’s garden? Is it better to coat seeds at the time of planting, or to water plants with a diluted solution?
I have pisolithus tinctorius growing in my yard, I don’t find them EVERY year, but I found two this year, one two years before that, all in different locations. What I did was I put water in a five gallon bucket and added a shovel full of soil along with some leaves/ compost like dry material (about 1 gallon in volume, I have a one gallon tupperware type juice pourer that I lost the lid to ages ago and now it stays out in the garden so I can dip it into the five gallon bucket when I mix up nutrients and and bacterial pest control for soil drenching) that would have gone onto the compost heap. Then I chopped up the mushroom and stuck it in the bucket with a lid over it and let it sit for at least one month out in our shed. Then I stirred it up well and used the water on all my vegetables. I don’t know if it was the mushroom tea or the mychorrizae that I bought from amazon, but my garden is amazing this year. I have beans, cucumbers, tomatoes and peppers in abundance.
I knew about mushroom tea for the garden years ago and had never tried it, but when I found the dog-turd mushroom in my yard I sent it away to get it positively id’ed (had NO CLUE what this weird looking thing was, especially because the form dried out but remained solid for two months after it formed just sitting there in the soil minding it’s business, unlike most mushrooms that literally dissolve within a week or so), then researched it-found out the cool stuff about it, so, I figured that would be a perfect fungus for mushroom tea for the garden. I imagine you can also coat the seeds or the seedling roots with the same solution at planting time.
Sorry so long, but I try to give reasons for each step I take when doing stuff for my garden when people ask, so yeah, a bit wordy.
TLDR; I personally use it as a mushroom tea after steeping it in a covered container for at least one month.
Reply