The comb tooth mushroom, Hericium coralloides (Scop.) Pers.
Classification
Kingdom Fungi
Phylum Basidiomycota
Class Basidiomycetes
Order Russulales
Family Hericiaceae
Genus Hericium
Synonyms
Clavaria madreporaeformis Retz.
Suppl. Scand. 1: 19 (1779)
Dryodon acicularis (Sacc.) Bourdot [as ‘aciculare‘]
Bull. trimest. Soc. mycol. Fr. 48: 221 (1932)
Dryodon coralloides (Scop.) P. Karst.
Meddn Soc. Fauna Flora fenn. 6: 15 (1881)
Dryodon coralloides var. crispus Cejp
(1928)
Friesites caput-ursi (Fr.) P. Karst.
Meddn Soc. Fauna Flora fenn. 5: 41 (1880)
Friesites coralloides (Scop.) P. Karst. [as ‘corallioides‘]
Meddn Soc. Fauna Flora fenn. 5: 41 (1880)
Hericium abietinum (Schrad.) Schleich.
Catalogus Plant. Helvetia: 57 (1821)
Hericium alpestre f. caput-ursi (Fr.) Nikol.
Acta Inst. Bot. Acad. Sci. USSR Plant. Crypt., Fasc. II 5: 337 (1950)
Hericium caput-ursi (Fr.) Corner
Bull. Br. Mus. nat. Hist., Bot. 1(7): 192 (1955)
Hericium laciniatum (Leers) Banker
Mem. Torrey bot. Club 12: 114 (1906)
Hericium ramosum (Bull.) Letell.
Hist. Champ. France (Paris): 43 (1826)
Hericium ramosum f. caput-ursi (Fr.) D. Hall & D.E. Stuntz
Mycologia 63: 1111 (1971)
Hericium reichii Opiz
Lotos 1: 256 (1851)
Hydnum abietinum Schrad.
Spicil. Fl. Germ. 1: 181 (1794)
Hydnum aciculare Sacc.
Michelia 2(no. 6): 154 (1880)
Hydnum caput-ursi Fr.
Monogr. Hymenomyc. Suec. (Upsaliae) 2(2): 278 (1863)
Hydnum coralloides Scop.
Fl. carniol., Edn 2 (Vienna) 2: 472 (1772)
Hydnum laciniatum Leers
Flora herbornensis: 276 (1775)
Hydnum novae-zelandiae Colenso
Proc. N.Z. Inst. 21: 79 (1889)
Hydnum ramosum Schwein.
Herbier de la France: 390 (1788)
Manina caput-ursi (Fr.) Banker
Mycologia 4(5): 277 (1912)
Manina coralloides (Scop.) Banker
Mycologia 4(5): 276 (1912)
Medusina coralloides (Scop.) Chevall.
Fl. Gén. Env. Paris 1: 279 (1826)
Merisma coralloides (Scop.) Spreng.
Syst. veg., Edn 16 4: 496 (1827)
Common names
Bear’s head tooth
Comb tooth
Conifer coral
Coral spine fungus
Coral tooth
Ästiger Stachelbart (German)

My name is Austin Collins.
I've dedicated my life to Mushrooms.
I believe Mushrooms are the best kept secret when it comes to health and well being.
For that reason, I would like to share a company with you that in my opinion makes the best mushroom products on the market.
The company is called Noomadic Herbals, my favorite supplement they make is called "Mushroom Total".
I take their products every day and they have helped me think better and have more energy. Give them a try.
-Austin
Description
Fruiting body: 8-30 cm diameter, fleshy, white at first, light brown or yellowish with age, a few main branches arising from the narrow base, every main branch sending forth numerous smaller branches, on which dense and crowded spines hang in rows.
Spines: cylindrical, pointed at apex, 0.5 – 1.5 cm long.
Spore print: white.
Spores: colorless, smooth, ellipsoid to subglobose, amyloid, hyaline, 5-8 x 4.5-6 µm.
Habitat: solitary or clustered clumps, usually on dead hardwood trees; saprobic; late summer and fall; common.
Odor: indistinct.
Edibility: edible.
Bioactive compounds
An early study on polysaccharides isolated from this mushroom revealed that it is a linear molecule containing only α-1,4 glucosidic linkages. Also, the polysaccharide differs from that of higher plants in that it consists only of short-chain amylose molecules (32 to 45 glucose units long), whereas in plants the polysaccharides may have chain lengths extending into the thousands, depending on the species (McCracken and Dodd, 1971).
Experiments using radiolabeled glucose suggest that the D-xyloside herical, isolated from cultures of H. coralloides, is thought to be a precursor in the synthesis cyathane-skeleton molecules, including erinacine E, discussed below. Herical is known to inhibit a large spectrum of fungi and bacteria and shows cytotoxic and hemolytic properties (Anke et al., 2002).
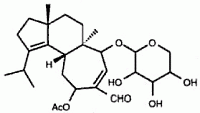
Medicinal properties
Nematicidal activity
A nematicidal fatty acid mixture was obtained from cultures of H. coralloides. This mixture, which contained primarily linoleic, oleic, and palmitic acids, showed toxic effects towards Caenorhabditis elegans (Stadler et al., 1994).
In 1998, a Pfizer research group isolated erinacin E (see structure below) from the fermentation broth of H. coralloides (Saito et al., 1998). Erinacin E is a highly selective agonist at the κ opiod receptor(IC50 of 0.8 µM compared with binding at the µ opiod receptor, IC50 of >200 mM). This molecule was earlier isolated from fruiting bodies of H. erinaceum, where it was reported to be a potent stimulator of nerve growth factor synthesis (Kawagishi et al., 1996). Stimulators of nerve growth factor synthesis are being investigated as potential medicines for degenerative neuronal disorders such as Alzheimer’s disease and peripheral nerve regeneration (Yamada et al., 1997). The total synthesis of erinacine E has recently been reported (Watanabe and Nakada, 2008).
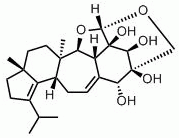
Links
Mushroom Expert
Fungi of Poland
Fungi on Wood
References
Anke T, Rabe U, Schu P, Eizenhofer T, Schrage M, Steglich W.
Studies on the biosynthesis of striatal-type diterpenoids and the biological activity of herical.
Z Naturforsch C. 2002 57(3-4):263-71.
Ginns J.
Hericium coralloides N. amer auct (Hericium americanum sp nov) and the European Hericium alpestre and Hericium coralloides.
Mycotaxon. 1984 20(1):39-43.
Hallenberg N.
Hericium coralloides and Hericium coralloides (basidiomycetes) in Europe.
Mycotaxon. 1983 18(1):181-9.
Kawagishi H, Shimada A, Hosokawa S, Mori H, Sakamoto H, Ishiguro Y, Sakemi S, Bordner J, Kojima N, Furukawa S.
Erinacines E, F, and G, stimulators of nerve growth factor (NGF)-synthesis, from the mycelia of Hericium erinaceum.
Tetra Lett. 1996 37(41):7399-402.
McCracken DA, Dodd JL.
Molecular structure of starch-type polysaccharides from Hericium ramosum and Hericium coralloides.
Science. 1971 174(7):419.
Pubmed
Saito T, Aoki F, Hirai H, Inagaki T, Matsunaga Y, Sakakibara T, Sakemi S, Suzuki Y, Watanabe S, Suga O, Sujaku T, Smogowicz AA, Truesdell SJ, Wong JW, Nagahisa A, Kojima Y, Kojima N.
Erinacine E as a kappa opioid receptor agonist and its new analogs from a basidiomycete, Hericium ramosum.
J Antibiot. 1998 51(11):983–90.
Pubmed
Stadler M, Mayer A, Anke H, Sterner O.
Fatty acids and other compounds with nematicidal activity from cultures of Basidiomycetes.
Planta Med. 1994 60(2):128-32.
Pubmed
Watanabe H, Nakada M.
Biomimetic total synthesis of (-)-Erinacine E
J Am Chem Soc. 2008 130(4):1150–1.
Yamada K, Nitta A, Hasegawa T, Fuji K, Hiramatsu M, Kameyama T, Furukawa Y, Hayashi K, Nabeshima T.
Orally active NGF synthesis stimulators: potential therapeutic agents in Alzheimer’s disease.
Behav Brain Res. 1997 83(1-2):117-22.
Pubmed




Hello,
We are mushroom growers from Western Australia. Are you able to tell me if there is a similarity between the medicinal benefits of Hericium colloroides and Hericium Erinaceaus
Regards
Graham Upson
Touchwood mushrooms
Hi,
Graham Upson – Yes they do and from researching i have found they have 3 extra products that also contain anti cancer properties 🙂 ,
Hi, Austin,
In your research, have you discovered whether H. Coralloides produces greater or lesser quantities of Erinacin E as compared to H. Erinaceum? My interest in growing lion’s mane is for cognitive health, so I would like to ask advice from an expert as to which strain would best serve this purpose.
Thanks,
Patrick Castrillo
Hericium erinaceus has much more research behind it.